NEAR INFRARED BIOIMAGING
Deep-tissue, high-contrast imaging at the in vivo level represents a critical diagnostic step
both at the preclinical and clinical level. In our group, we push the limits of optical imaging approaches employing fluorescence nanoparticles operating in the
so-called biological windows. These are spectral ranges located in the infrared region (750-1800 nm),
in which tissues are partially transparent to the electromagnetic radiation.
We have been pioneers in the development of cost-effective, highly versatile experimental
set-ups for in vivo infrared fluorescence imaging. We also develop suitable contrast agents
composed of infrared-emitting semiconductor nanocrystals and rare earth-doped nanoparticles.
The imaging techniques we employ at nanoBIG include hyperspectral in vivo imaging,
in vivo lifetime imaging, and
subtissue fluorescence-assisted thermal videorecording (see figure).
Aside from purely imaging purposes, we employ the nanomaterials we develop for theranostics (combined therapy and diagnostics).

NANOTHERMOMETRY AND HYPERTHERMIA
Monitoring of the thermal state of biological systems can afford information about
processes occurring down to the single-cell level. On the other hand, in the context of thermal therapies (i.e., magnetic hyperthermia and photothermal therapy),
real-time temperature monitoring allows on-the-fly adjustments of the therapeutic parameters.
To that end, in the toolbox of methods for non-invasive
thermometry, semiconductor nanocrystals have emerged as efficient multifunctional
fluorescent nano-thermometers capable of simultaneously generating intracellular imaging, as well as
acting as optical thermal indicators. For example, they have been used for dynamical thermal monitoring of
single cells in the proximity of heat sources or during externally induced thermogenesis processes.
Compared to other methods, semiconductor nanocrystals-based thermometry
offer superior performance such as sub-micrometric resolutions,
high contrast, good biocompatibility, excellent temperature resolution (of about 0.2 °C)
and possibility of selective targeting via surface functionalization.

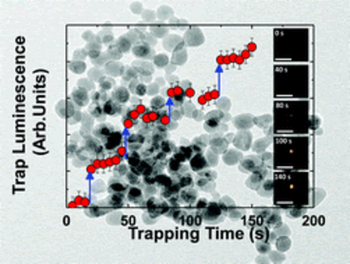
OPTICAL TRAPPING
Optical tweezers
are scientific instruments that use a highly
focused laser beam to create an attractive or repulsive force - typically in the
order of picoNewtons, and dependent upon the refractive index mismatch - to physically "grab" and move
dielectric objects at the micro- and nanoscale.
Optical tweezers have been particularly useful
for the study of a variety of biological systems in recent years, as they can be used
to immobilize and manipulate small objects (e.g., a nanoparticle) into the system of interest (e.g., a cell).
Single-beam optical tweezers were initially introduced by Ashkin et al.: Therein, a single laser beam is focused through a high numerical
aperture objective lens to generate a three-dimensional optical trap by exerting
a radiation force on small objects.
Since then, optical tweezers have become calibrated tools to immobilize, orient, and transport sub-micrometric particles.
The nanometer positioning ability along with sub-picoNewton force resolution have turned this approach into
a valuable tool in biology. However, direct trapping and manipulation of smaller particles still
represents a challenge, as they are substantially harder to trap.
OPTICAL COHERENCE TOMOGRAPHY
Optical coherence tomography (OCT) is based on low-coherence interferometry,
typically employing near-infrared light, which enables deep-tissue imaging with typical resolutions of
~10-20 µm and reaching penetration depths as large as 3 mm in nontransparent media.
In our lab, we host an intracoronary OCT (IC-OCT) system used in clinical practice at the Interventional
Cardiology Unit of the Hospital Universitario La Princesa of Madrid (Dragonfly OPTIS Imaging Catheter St Jude Medical)
as well as an open OCT system (Telesto Thorlabs OCT system).
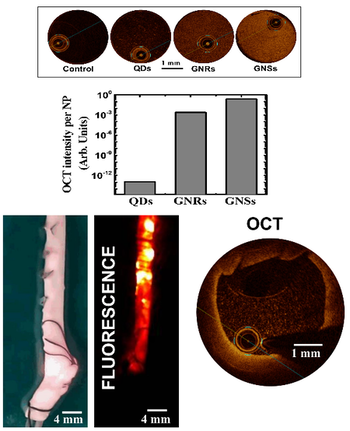
We can harness the light scattering properties of plasmonic gold-based nanoparticles and PbS quantum dots
to obtain positive OCT contrast (see image). In the case of near-infrared emitting PbS quantum dots, the simultaneous use of the
OCT setup and an infrared camera enables OCT and two-dimensional luminescence
imaging of arteries (see image). The use of highly absorbingplasmonic nanomaterials (e.g., CuS nanoparticles) allows instead to generate dark contrast
in OCT images, which is envisaged to be of utility in the precise localization of atheromata.
SPECTROSCOPY AND FLUORESCENCE IMAGING OF PHOTONIC DEVICES
Confocal fluorescence imaging has demonstrated to be
a powerful technique for the characterization and understanding of
a great variety of micro- and nano-photonic devices.
NanoBIG has worked extensively on the application of confocal
fluorescence imaging for the acquisition of fluorescence images of ultrafast
laser-inscribed photonic devices in laser gain media.
These images have been pivotal to understand the role played by the different
micro-structural modifications induced by ultra-intense laser pulses (stress, damage, disorder)
in the performance of diffeent structures ranging from waveguides to photonic crystals.



Collaborations
- Università degli Studi di Verona (Italy) Prof. Marco Bettinelli
- Università degli Studi di Verona (Italy) Prof. Adolfo Speghin
- Concordia University (Quebec, Canada) Prof. John A. Capobianco
- Institut National de la Recherche Scientifique (Quebec, Canada) Prof. Fiorenzo Vetrone
- University of Ottawa (Ottawa, Ontario, Canada) Prof. Eva Hemmer
- Swinburne University of Technology (Melbourne, Australia) Prof. Min Gu
- Universidad de Sonora (México) Prof. Marcelino Barboza Flores
- Universidade Federal de Alagoas (Brazil) Prof. Carlos Jacinto
- Heriot-Watt University (Scotland, UK) Prof. Ajoy K. Kar
- University of St. Andrews (Scotland, UK) Prof. Kishan Dholakia
- National University of Singapore (Singapore) Prof. Xiaogang Liu
- Virginia Tech (USA) Prof. Chen Lu
- Université de Lyon (France) Prof. Georges Boulon
- Shandong University (China) Prof. Feng Chen
- University of Pittsburgh (USA) Prof. Kevin Chen
- Universidad Nacional Autónoma de México (Mexico) Prof. G. Hirata
- Aveiro Institute of Materials, Universidade de Aveiro (Portugal) Prof. Luis Dias Carlos
- Vinca Institute of Nuclear Sciences, University of Belgrade (Serbia) Prof. Miroslav Dramicanin
- Harbin Institute of Technology (China) Prof. Guanying Chen
- Polish Academy of Sciences in Wrocław (Poland) Prof. Artur Bednarkiewicz
- IRCP CNRS Chimie Paristech (France) Prof. Bruno Viana
- Tokio University (Japan) Prof. Kohei Soga
- University of Tartu (Estonia) Prof. Yurii Orlovskii
- University of Latvia (Latvia) Prof. Krisjanis Smits
- Université Paris-Sud Prof. Niko Hildebrandt
